RINGTOWN -- An estimated 1,000 women around the country have gone online to blame the Essure birth control device for their pain. Some said their problems did not surface until two or three years after the device was implanted.
Shortly after giving birth to her fifth child, Lisa Conti of Ringtown in Schuylkill County decided that was enough.
Her doctor recommended Essure implants for birth control.
"He told me there would be minimum pain afterwards, that if I did get anything, it would be mild cramping," said Conti.
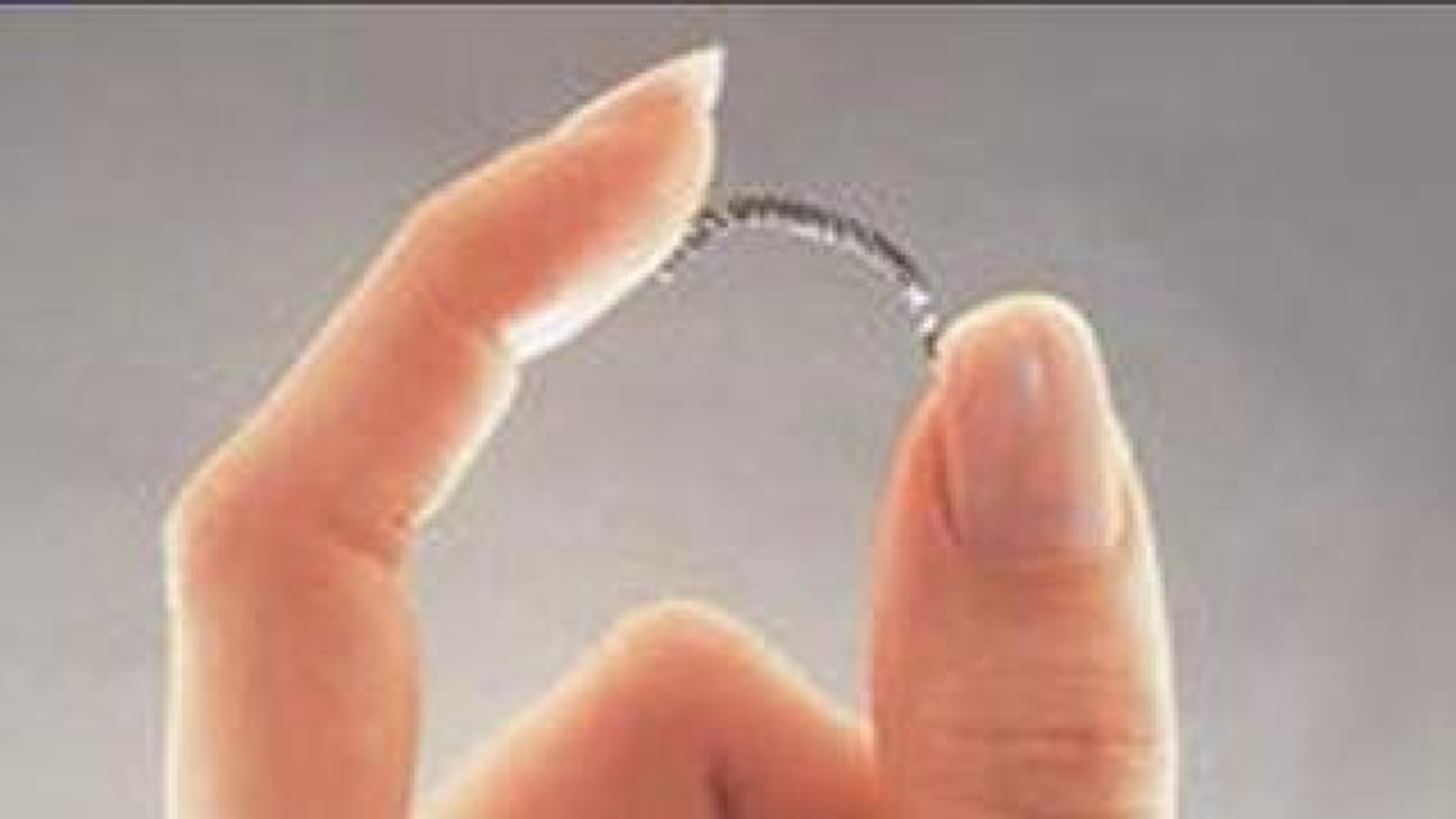
Essure implants are small, metal coils a doctor inserts in a woman's fallopian tubes, and within three months, enough scar tissue forms to block the tubes and prevent pregnancy.
After Lisa had Essure implants in 2009, she experienced minor pain, but after two years...
"I`d get sharp pains in my sides. Shooting down my legs," said Lisa. "My legs would go numb, and when I`d get a pain in my legs, I`d just drop to the floor."
At one point late last year, Lisa Conti`s pain was so unbearable, she had a friend drive her to the Emergency Room at the Schuylkill Medical Center in Pottsville. She told the doctor, she thought the Essure device was the source of her pain.
Lisa said the ER doctor on duty never heard of Essure.
The US Food and Drug Administration, which approved the implants in 2002 considers Essure safe.
An agency spokeswoman claims 750,000 women have been implanted with Essure coils, and only 597 women or their doctors have reported complications.
A well-known leading consumer advocate claims that number is too low.
"I had several hundred women coming to me," said Erin Brockovich.
From her office in Los Angeles, Brockvich told us she has received emails from more than 1,000 women reporting problems.
She said they range from unwanted pregnancies, to internal bleeding after an Essure coil dislodged.
Brockovich said the women cannot sue Essure's manufacturer Bayer because the FDA approved the device.
So she's posting some of these women's stories her web page, and collecting petitions to bring to congress.
"Just take it off the market, take a look at what went wrong, let's make it right before we put it back out there, and help those who have been harmed," said Brockovich.
Geisinger Medical Center, Danville OB/GYN Dr. Stanley Frye said despite the controversy, Essure remains a viable form of birth control.
"It's about as close to 100% effective as you can get," said Dr. Frye who adds Essure costs far less than a hysterectomy, avoids the risk of surgery, and has other benefits for women.
"They can return to work the following day, return to normal functions the following day, compared to other modalities that could be a week at a time," added Dr. Frye.
Lisa Conti hopes women consider her story, and those of others before considering Essure.
"The worst part is not feeling like myself and being there for my kids like I should be, like I was before in the past," she said.
In a statement, the Bayer Company, which manufactures Essure said:
"At Bayer, we care about patients and take the safety of our products very seriously. We are saddened to hear of any serious health condition affecting a patient using one of our products, irrespective of the cause. Essure was approved by the FDA in 2002, and has a well-documented benefit-risk profile, with over 400 peer-reviewed publications and abstracts supporting Essure’s safety, efficacy (desired result), and cost-effectiveness. Approximately 750,000 women worldwide rely upon the Essure procedure for permanent birth control. A recent practice bulletin issued by the American College of Obstetricians and Gynecologists (ACOG) has recognized that hysteroscopic tubal occlusion for sterilization has high efficacy and low procedure-related risk, cost, and resource requirements.
No form of birth control is without risk or should be considered appropriate for every woman. It is important that women discuss the risks and benefits of any birth control option with their physicians."